-
Regardless of the field, performed all human subject research on campus - Types of human subject research
Human subject research
- Observational research
: Observing, recording, analysis in behavior of subjects - Survey research
: Derivation of specific information via survey or interview in subjects or subject group - Experimental Research
: This research is to look at the results after experimental situation of subjects or deliberately
changing environmental conditions for verifying the specific hypothesis or acquiring novel information.
Experiments used for the purpose of medical research are therapeutic research (application of invented new treatment to patients) and non-therapeutic research (performing for scholarly purpose without the nature of treatment)
Human materials research
: researching tissues, cells, bloods, and etc. - Observational research
-
-
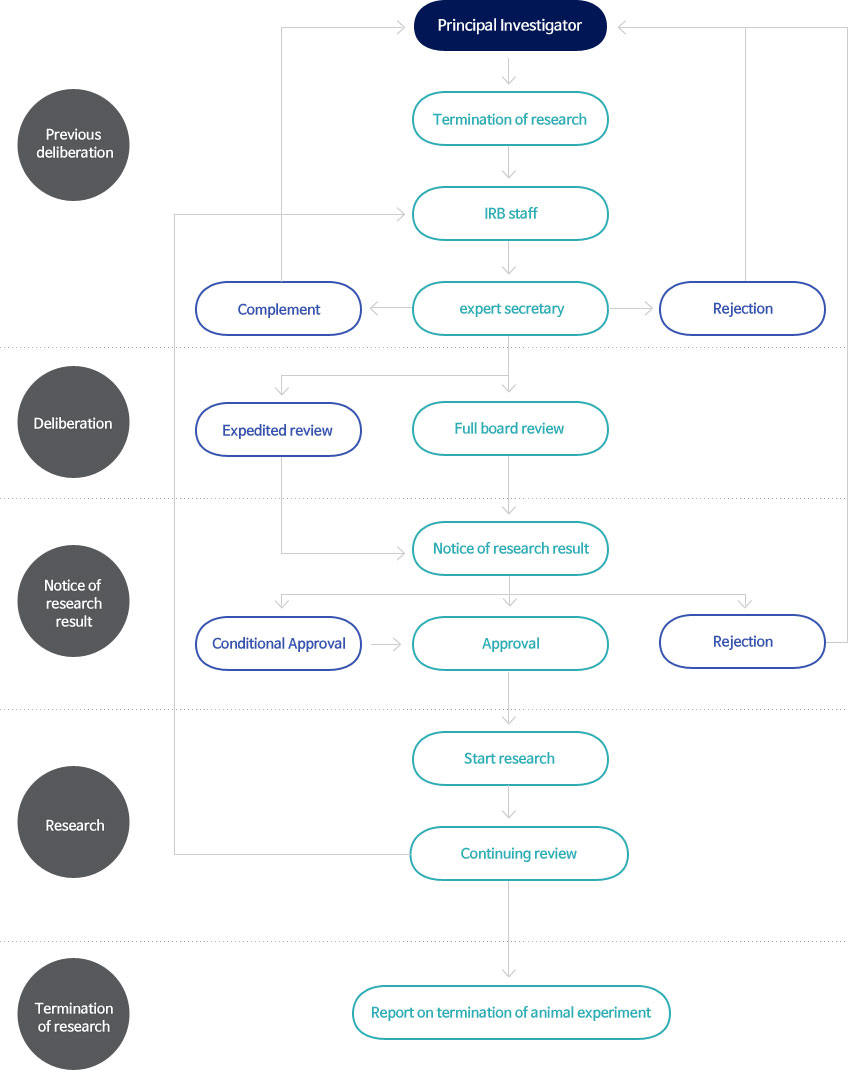
How to submit
R-Bay System : https://r-bay.co.kr
Documents to be submitted
1. Initial protocol review (planned protocols reviewed for the first time by IRB)
1) Common documents
- [IRB_1] Protocol review application
- [IRB_1-1,2,3] Research proposal
2) Additional documents
- When receiving resources from outside UNIST to be used in research : IRB approval certificate from resource providing institute
- For clinical trials: recruit subjects, survey sheet
- [IRB_6] Informed consent form
2. Protocol revision review
- [IRB_3] Protocol revision application
- [IRB_3-1] Change comparison table
- Other documentary evidence for supporting what has been stated in the request (only if necessary)
3. Continuing review
- [IRB_4] Report_on interim of research
- Other documentary evidence for supporting what has been stated in the request (only if necessary)
IRB schedule
1. Full board review
- held the second week of each month. (about protocols received of last months)
2. Expedited review
- may be submitted anytime and be reviewed by IRB.
Contact
Yoonjin Lee
- Tel. 052-217-5214
- Fax. 052-217-5219
- e_mail : leeyj0926@unist.ac.kr
- Office : Stem cell research center(105)-109